Welcome to Union-agrochemCrop Protection Technology (Shanghai) Co.,ltd
Toggle Navigation
|
Glufosinate-ammonium IUPAC name ammonium (S)-2-amino-4-[hydroxy(methyl)phosphinoyl]butyrate; ammonium DL-homoalanin-4-yl(methyl)phosphinate 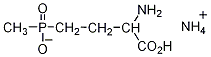
|
Chemical Abstracts name ammonium (±)-2-amino-4-(hydroxymethylphosphinyl)butanoate CAS RN [77182–82–2] unstated stereochemistry EC no 278–636–5 Development codes AE F039866 (AgrEvo); Hoe 039866 (Hoechst) Smiles code [NH4+].CP(=O)([O-])CCC(N)C(=O)O |
|
PHYSICAL CHEMISTRY |
|
Mol. wt. 198.2 M.f. C5H15N2O4P Form Crystalline solid, with a slightly pungent odour. M.p. 215 °C V.p. <3.1 × 10-2 mPa (50 °C) Kow logP <0.1 (pH 7, 22 °C) S.g./density 1.4 (20 °C) Solubility In water >500 g/l (pH 5–9, 20 °C). In acetone 0.16, ethanol 0.65, ethyl acetate 0.14, toluene 0.14, hexane 0.2 (all in g/l, 20 °C). Stability Stable to light and to hydrolysis at pH 5, 7 and 9. |
|
APPLICATIONS |
|
|
|
Biochemistry Glutamine synthetase inhibitor; leads to accumulation of ammonium ions, and inhibition of photosynthesis. Mode of action Non-selective contact herbicide with some systemic action. Translocation occurs only within leaves, predominantly from the leaf base to the leaf tip. |
|
Uses Glufosinate-ammonium is used for control of a wide range of annual and perennial broad-leaved weeds and grasses in fruit orchards, vineyards, rubber and oil palm plantations, ornamental trees and bushes, non-crop land, and pre-emergence in vegetables. Also used as a desiccant in potatoes, sunflowers, etc. For control of annual and perennial weeds and grasses in glufosinate-tolerant crops (oilseed rape, maize, soya beans, sugar beet) developed through gene technology. Applied at 0.4–1.5 kg/ha. |
|
Formulation types SL. Compatibility Compatible with diuron, simazine, MCPA, and some other herbicides. |
|
ANALYSIS |
|
Product analysis by hplc with uv determination (CIPAC Handbook, 1995, G, 89–93). Residues in plants or in soil by gc/FPD (Resid. Anal. Methods; Environ. Chem. Methods); see also Pestic. Anal. Man., II, 180.473. Details available from Bayer CropScience. |
|
TOXICOLOGICAL & ENVIRONMENTAL REVIEWS |
|
JMPR Mtg. 86 (1999); JMPR Evaln. I 72 (1994), 84 (1998), 87 (1999); JMPR Evaln. II 64 (1991), 88 (1999). EU Rev. Rep. SANCO/10453/2006; EFSA Sci. Rep. (2005) 27, 1–81. E. Ebert et al., Fd. Chem. Toxic., 1990, 28(5), 339–349; R. Hack et al., ibid., 1994, 32(5), 461–470. 91/414/EC Annex I status Included, 2007/25/EC. |
|
MAMMALIAN TOXICOLOGY |
|
Oral Acute oral LD50 for male rats 2000, female rats 1620, male mice 431, female mice 416, dogs 200–400 mg/kg. Skin and eye Acute percutaneous LD50 for male rats >4000, female rats c. 4000 mg/kg. Not a skin or eye irritant. Inhalation LC50 (4 h) for male rats 1.26, female rats 2.60 mg/l air (dust); for rats >0.62 mg/l air (aerosol). NOEL (2 y) for rats 2 mg/kg b.w. daily. ADI/RfD (JMPR) 0.02 mg/kg b.w. (for glufosinate-ammonium, N-acetylglufosinate and 3-methylphosphinico-propionic acid, alone, or in combination) [1999]; (EC) 0.021 mg/kg b.w. [2007]; (EPA) lowest aRfD 0.063, cRfD 0.02 mg/kg b.w. [1993]. Other No teratogenic, carcinogenic, mutagenic or neurotoxic effects have been observed. Toxicity Class WHO (a.i.) III; EPA (formulation) III EC classification R60| R63| Xn; R20/21/22, R48/20/22 |
|
ECOTOXICOLOGY |
|
Birds Dietary LC50 (8 d) for Japanese quail >5000 mg/kg. Fish LC50 (96 h) for rainbow trout 710, carp, bluegill sunfish, golden orfe >1000 mg/l. Daphnia LC50 (48 h) 560–1000 mg/l. Algae LD50 for Scenedesmus subspicatus ≥1000, Selenastrum capricornutum 37 mg/l. Bees Not hazardous to bees; LD50 >100 μg/bee. Worms LD50 for earthworms >1000 mg/kg soil. Other beneficial spp. Not toxic to beneficial arthropods. |
|
ENVIRONMENTAL FATE |
|
Animals Rapidly excreted, predominantly via faeces (90%). The principal metabolite is 3-(methyl)phosphinoylpropionic acid. A further faecal metabolite is N-acetylglufosinate, formed by intestinal micro-organisms. Plants Non-selective use: only the metabolite, 3-(methyl)phosphinoylpropionic acid (3-MPP), is taken up in traces from the soil. Desiccation: most of the residues consist of parent glufosinate-ammonium, with minor amounts of metabolite 3-MPP. Selective use: the principal metabolite is N-acetylglufosinate, with lesser amounts of parent and 3-MPP. Soil/Environment Rapidly degraded in surface levels of soil, and in water. Because of polarity, it and its metabolites do not bioaccumulate. Metabolism in soil and water reviewed (E. Dorn et al., Z. Pflanzenkr. Pflanzenschutz, 1992, Sonderheft XIII, pp. 459–468). Degraded to 3-(methyl)phosphinoylpropionic acid and 2-(methyl)phosphinoylacetic acid, and ultimately to CO2 and bound residues. In soil, DT50 3–10 d (lab.), 7–20 d (field); DT90 10–30 d (lab.); DT50 of metabolites 7–19 d (lab.). DT50 in water c. 2–30 d. Lysimeter studies and model calculations show that neither a.i. nor metabolites leach into groundwater; this appears to be due to rapid degradation, and adsorption to certain soil elements. Adsorption is more correlated with clay content than organic matter, Kclay 2–115, Koc 10–1230 (A. Zumdick et al., Proc. 9th IUPAC Int. Congr. Pestic. Chem., London, 1998, 2, 6A-023; idem, ibid., 6D-034). |